Advances in
Clinical Care
We strive to meet the needs of our patients by bringing them the benefits of our breakthroughs and providing them with superior, state-of-the-art care.
Improved outcomes in stem cell transplants

UChicago Medicine has the highest one-year survival rate (80%) in Illinois for adults undergoing blood and bone marrow stem cell transplants, according to the latest statistics from the Center for International Blood & Marrow Transplant Research (CIBMTR). The national average was 76.5%. CIBMTR said UChicago Medicine’s outcomes are statistically in line with expectations based on the illnesses of the patients it treated. UChicago Medicine typically cares for particularly ill patients whose prognoses are grim before their transplants. Based on the illnesses of UChicago Medicine’s patients, CIBMTR predicted the academic medical center would have a 76.4% survival rate. Its 80% adult survival rate meant the organization exceeded its predicted performance by 3.6%. No other Illinois stem cell transplant program exceeded its predicted survival rate by a higher margin.
Learn more“These outcomes are a direct reflection
of the tremendous teamwork and
efforts by all members of our
transplant and cell therapy program.”

Michael R. Bishop, MD
Professor of Medicine and Director of the Jonas Center
Help navigating the cancer journey
In 2022, UChicago Medicine received a $300,000, multiyear grant from the American Cancer Society for a program to enhance oncology patient navigation and address barriers to individualized, timely and equitable care for cancer patients and their families. Navigation is a crucial component of cancer care, from prevention through treatment and survivorship. By providing individualized assistance to patients, families and caregivers, navigation ensures high-quality health and psychosocial care, creating positive health outcomes for patients.
Learn moreA cancer center of the future

UChicago Medicine announced its plans to build Chicago’s first freestanding clinical cancer center. The $815 million, 575,000-square-foot facility dedicated to cancer care is one of the largest investments made by the academic health system for patients and the community. A significant portion of the planning and design will focus on the patient and family experience, including making sure all services throughout the care journey are in the same location and creating a healing and stress-reducing environment. The cancer center, which includes inpatient and outpatient care, will have a focus on prevention and early detection of cancer and be a hub for research into the more aggressive forms of cancer that affect residents on the South Side and in many other communities of color across the country. Pending regulatory approval, construction of the new facility will begin in 2023 and it will open to patients in 2027.
Learn moreTheranostics enables precise diagnosis and treatment of cancers
Theranostics — the word is a blend of "therapy" and "diagnostics" — involves the use of radioactive isotopes to diagnose and treat cancer. Physicians have been using radioactive isotopes like technetium-99 and strontium-89 to diagnose and treat cancer patients for years. Theranostics, however, uses radiotracers made up of two components: a biological component engineered to latch on to a specific protein on a cancer cell, and interchangeable radioactive isotopes that can alternatively make that cell visible on a PET scan and then kill it. With the only academic health system cyclotron in Illinois capable of producing novel radioactive agents, and in collaboration with Argonne National Laboratory, UChicago Medicine scientists are working to develop better theranostics that build off of those currently or soon available.
Learn morePATIENT SPOTLIGHT
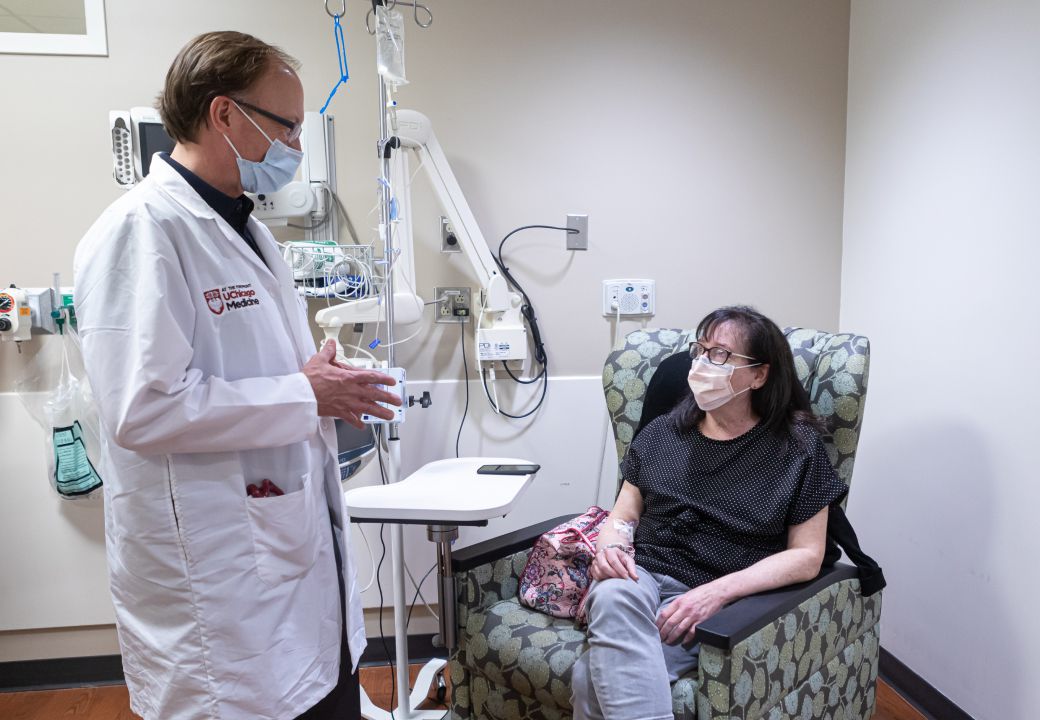
A groundbreaking new drug for rare eye cancer
Tebentafusp-tebn was approved by the FDA in January 2022. However, it’s been helping University of Chicago Medicine cancer patient Kathy Huyser for several years. Huyser was part of a clinical trial at UChicago Medicine of tebentafusp-tebn as a treatment for her uveal melanoma, a rare type of cancer that forms in the back of the eye. It can be hard to detect, giving the cancer an opportunity to spread throughout the body. Participants, including Huyser, received weekly intravenous (IV) infusions and blood tests, and CT scans every 12 weeks. While the drug did not help every patient in the trial, it proved to be a viable tool in fighting uveal melanoma. In Huyser’s case, it shrunk her tumors and has kept her cancer under control for four years. Because UChicago Medicine was one of just a handful of hospitals in the United States to participate in this clinical trial, it is now one of the first centers nationwide offering tebentafusp-tebn to uveal melanoma patients.
Read full articleSeeing traction in push to lower cancer drug costs
Mark Ratain, MD, Leon O. Jacobson Professor of Medicine, has spent nearly two decades advocating for changes that he believes will significantly lower the cost of oncology drugs. His work influenced a bill that would establish an interagency task force between the patent office and the FDA. The task force would make sharing information across the agencies easier and allow the FDA to provide technical assistance on prescription drug patents. The bill was unanimously supported by the Senate Judiciary Committee on July 28, 2022, and will go to a full Senate vote. Ratain said that while policies only budge the needle on cancer drug costs, it’s at least a move in the right direction.
Learn more